- Nuclear Atom Picture
- Nuclear Atom Physics
- Structure Of The Nuclear Atom
- Nuclear Atom Splitting
- Nuclear Atom Curtains Panels
Atom is an award-winning app and website that makes it easy to find new movie releases playing in theaters near you. Buy tickets for the latest movie showtimes and hot movies out this week plus special movie events in theaters. The single most important characteristic of an atom is its atomic number (usually denoted by the letter Z), which is defined as the number of units of positive charge (protons) in the nucleus. For example, if an atom has a Z of 6, it is carbon, while a Z of 92 corresponds to uranium. Nuclear reactors are the heart of a nuclear power plant. They contain and control nuclear chain reactions that produce heat through a physical process called fission. That heat is used to make steam that spins a turbine to create electricity. With more than 440 commercial reactors worldwide.
- Atomic model
- Basic properties
- The electron
- The nucleus
- Development of atomic theory
- The beginnings of modern atomic theory
- Studies of the properties of atoms
- Models of atomic structure
- Advances in nuclear and subatomic physics
Dungeon of the endless™ - crystal edition download for mac. Our editors will review what you’ve submitted and determine whether to revise the article.
Join Britannica's Publishing Partner Program and our community of experts to gain a global audience for your work!Atom, smallest unit into which matter can be divided without the release of electrically charged particles. It also is the smallest unit of matter that has the characteristic properties of a chemical element. As such, the atom is the basic building block of chemistry.
Most of the atom is empty space. The rest consists of a positively charged nucleus of protons and neutrons surrounded by a cloud of negatively charged electrons. The nucleus is small and dense compared with the electrons, which are the lightest charged particles in nature. Electrons are attracted to any positive charge by their electric force; in an atom, electric forces bind the electrons to the nucleus.
Because of the nature of quantum mechanics, no single image has been entirely satisfactory at visualizing the atom’s various characteristics, which thus forces physicists to use complementary pictures of the atom to explain different properties. In some respects, the electrons in an atom behave like particles orbiting the nucleus. In others, the electrons behave like waves frozen in position around the nucleus. Such wave patterns, called orbitals, describe the distribution of individual electrons. The behaviour of an atom is strongly influenced by these orbital properties, and its chemical properties are determined by orbital groupings known as shells.
This article opens with a broad overview of the fundamental properties of the atom and its constituent particles and forces. Following this overview is a historical survey of the most influential concepts about the atom that have been formulated through the centuries. For additional information pertaining to nuclear structure and elementary particles, seesubatomic particles.
Atomic model
Most matter consists of an agglomeration of molecules, which can be separated relatively easily. Molecules, in turn, are composed of atoms joined by chemical bonds that are more difficult to break. Each individual atom consists of smaller particles—namely, electrons and nuclei. These particles are electrically charged, and the electric forces on the charge are responsible for holding the atom together. Attempts to separate these smaller constituent particles require ever-increasing amounts of energy and result in the creation of new subatomic particles, many of which are charged.
As noted in the introduction to this article, an atom consists largely of empty space. The nucleus is the positively charged centre of an atom and contains most of its mass. It is composed of protons, which have a positive charge, and neutrons, which have no charge. Protons, neutrons, and the electrons surrounding them are long-lived particles present in all ordinary, naturally occurring atoms. Other subatomic particles may be found in association with these three types of particles. They can be created only with the addition of enormous amounts of energy, however, and are very short-lived.
All atoms are roughly the same size, whether they have 3 or 90 electrons. Approximately 50 million atoms of solid matter lined up in a row would measure 1 cm (0.4 inch). A convenient unit of length for measuring atomic sizes is the angstrom (Å), defined as 10−10 metre. The radius of an atom measures 1–2 Å. Compared with the overall size of the atom, the nucleus is even more minute. It is in the same proportion to the atom as a marble is to a football field. In volume the nucleus takes up only 10−14 metres of the space in the atom—i.e., 1 part in 100,000. A convenient unit of length for measuring nuclear sizes is the femtometre (fm), which equals 10−15 metre. The diameter of a nucleus depends on the number of particles it contains and ranges from about 4 fm for a light nucleus such as carbon to 15 fm for a heavy nucleus such as lead. In spite of the small size of the nucleus, virtually all the mass of the atom is concentrated there. The protons are massive, positively charged particles, whereas the neutrons have no charge and are slightly more massive than the protons. The fact that nuclei can have anywhere from 1 to nearly 300 protons and neutrons accounts for their wide variation in mass. The lightest nucleus, that of hydrogen, is 1,836 times more massive than an electron, while heavy nuclei are nearly 500,000 times more massive.
Basic properties
Atomic number
The single most important characteristic of an atom is its atomic number (usually denoted by the letter Z), which is defined as the number of units of positive charge (protons) in the nucleus. For example, if an atom has a Z of 6, it is carbon, while a Z of 92 corresponds to uranium. A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance. Since it is the electrons that determine how one atom interacts with another, in the end it is the number of protons in the nucleus that determines the chemical properties of an atom.
- key people
- related topics
The Nuclear Atom
Atomic Number
The radiations produced by uranium and thorium were quite feeble, anddifficult to work with. This situation was corrected by Mme. Curie.Investigating the radioactivity of uranium minerals, she found some samples ofore of low uranium content that were nevertheless intensely radioactive - moreso than if they had been pure uranium.
She reached the conclusion that the ore must contain some element other thanuranium that was radioactive. Since she knew all the components of the ore thatwere present in significant amounts, and since all were known to benon-radioactive, the unknown element could be present only in very smallquantities and must be extremely radioactive indeed.
During 1898, she and her husband slaved away over quantities of ore, tryingto concentrate the radioactivity and isolate the new element. In July, one newelement was located and named polonium, after Mme. Curie's nativePoland, and, in December, a second new element, radium.
Radium was extremely radioactive, giving off radiations in 300,000 times thequantity that the same weight of uranium did. Furthermore, it was very rare. Out of tons of ore, the Curies managed to obtain only about 1/300th of an ounceof radium.
Other strongly radioactive elements were discovered in tiny traces. In 1899,the French chemist Andre Louis Debierne (1874-1949)discovered actinium. In 1900, the German physicist Friedrich ErnstDorn (1848-1916) found a radioactive gas which eventuallyreceived the name radon. It was one of the inert gases, fitting belowxenon in the periodic table. Finally, in 1917, the German chemist OttoHahn (1879-1968) and Lise Meitner(1878-1968) discoveredprotactinium.
Experimenters could use these rare but extremely radioactive elements in 'particleguns'. Lead absorbs the radiation. If a bit of material containing one ofthese elements is placed in a lead-lined box with a hole in it, almost all theparticles that come flying off are absorbed by the lead. Some, however, maketheir way through the hole to compose a tin stream of very many, very energeticparticles which can be directed at some target.
It was Rutherford who used such 'particle guns' most effectively.Beginning in 1906, he bombarded thin sheets of metal (such as gold) withspeeding alpha particles. Most of the alpha particles passed clear through,unaffected and undiverted, recording themselves on the photographic platebehind. There were, however, some particles that were scattered - even throughlarge angles.
Since the gold foil that served as target was two thousand atoms thick, andsince most alpha particles passed through untouched, it would seem that theatoms were mostly empty space. Since some alpha particles were deflectedsharply, it meant that somewhere in the atom must be a massive, positivelycharged region, capable of turning back the positively charged alpha particle.
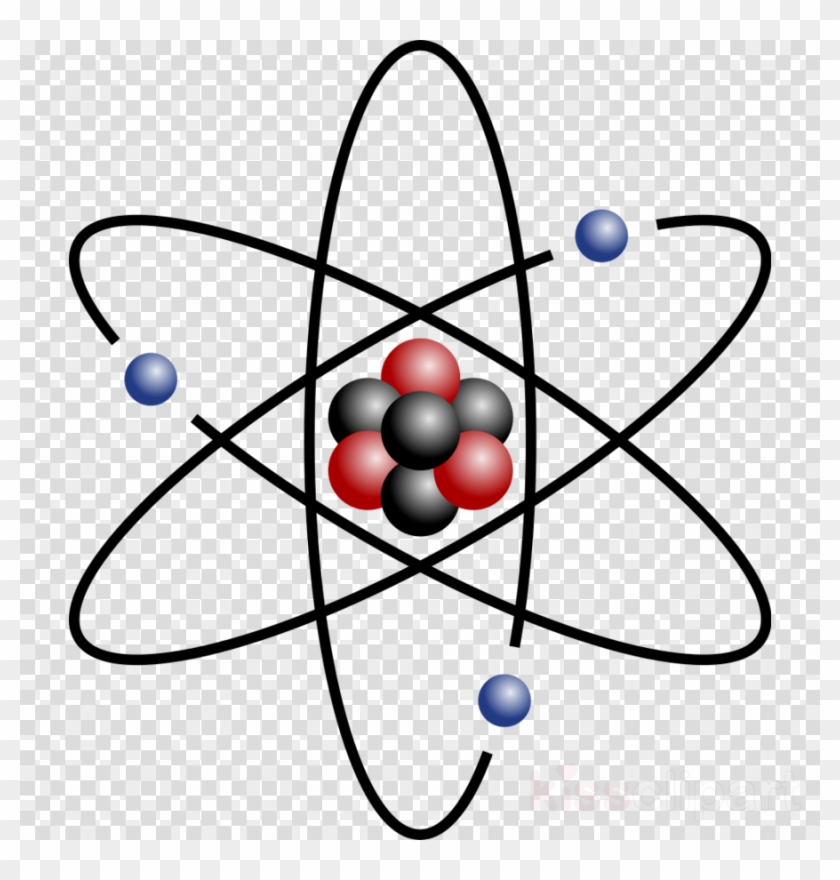
Rutherford therefore evolved the theory of the nuclear atom. Theatom, he decided, contains a very tiny nucleus at its center, which ispositively charged and which contains all the the protons (and, it was laterdiscovered, the neutrons, too) of the atom. The atomic nucleus has tobe very tiny in order to account for the very small fraction of the alphaparticles that were deflected, but it must also contain virtually all the massof the atom.
In the outer regions of the atom are the negatively charged electrons, whichare too light to interpose an important barrier to the passage of the alphaparticles. Although the protons and alpha particles are as massive as atoms,they are actually bare atomic nuclei. They take up so little room in comparisonwith the atom that they, too, despite their large mass, may be consideredsubatomic particles.
Rutherford's nuclear atom lent a new subtlety to the question of theindivisibility of the atom. The central nucleus, which was the heart of theatom, was surrounded and protected by a cloud of electrons. It remaineduntouched and intact through all chemical changes. It was this seemingpermanence of the nucleus that led all experimental evidence prior to the 1890'sto appear to suggest the notion of an indivisible atom.
However, the atom did undergo one type of change in ordinary chemicalreactions. Much of the electron cloud remained intact, but not all. Someelectrons could be removed from the 'surface' of the atom, or added tothat surface. In this way, the problem of ions, which had puzzled threegenerations of chemists, was finally solved.
If the nuclear atom is accepted, the next question is: How does the nuclearatom of one element differ from that of another?
Since Dalton's time, different atoms had been known to differ in mass, buthow is this difference reflected in the subatomic particles making up thenuclear atom?
The beginnings of an answer came through a study of x-rays. The Germanphysicist Max Theodor Felix von Laue (1879-1960) began, in1909, to bombard crystals with x-rays. These classic experiments establishedtwo vital facts: Crystals consist of atoms arranged in a geometrical structureof regular layers, and these layers scatter x-rays in a set pattern. From themanner in which the x-rays are scattered (or diffracted), the size of the tinywaves (wavelength) making up the x-rays can be determined.
Next, the English physicist Charles Glover Barkla(1877-1944) found, in 1911, that when x-rays are scattered by particularelements, they produce beams of x-rays that penetrate matter by characteristicamounts. Each element gives rise to a particular set of characteristicx-rays. Another English physicist, Henry Gwyn-Jeffreys Moseley(1887-1915), used Laue's method to determine the wavelengths of thesecharacteristic x-rays. He found, in 1913, that the wavelength of these x-raysdecreased smoothly with the increasing atomic weight of the elements emittingthem. This inverse relationship, Moseley argued, depended on the size of thepositive charge on the nucleus of the atom. The larger the charge, the shorterthe wavelength of the characteristic x-rays.
From the wavelength, in fact, it was possible to calculate what the chargemust be for the atoms of any particular element. Thus, as was eventually shown,hydrogen had a nuclear charge of +1, helium of +2, lithium of +3, and so on allthe way up to +92 for uranium. (These numbers are based on a standard accordingto which the charge on a proton is arbitrarily set equal to +1, and that on anelectron to -1.)
The size of the nuclear charge is called the atomic number. For thefirst time it was understood that when Mendeleev had arranged his elements inorder of what was taken to be atomic weight, he really was arranging them inorder of atomic number. In the couple of cases in which he had placed the moremassive atoms ahead of less massive ones, the less massive one nevertheless hadthe larger atomic number for reasons which will shortly be discussed.
Now it was finally possible to replace Boyle's operational definition of anelement, as a substance that could not be broken down into simpler substances,with a structural definition. The twentieth-century definition of an elementwould be: An element is a substance consisting of atoms that all possess anidentical and characteristic atomic number.
For the first time it became possible to predict exactly how many elementsremained to be discovered. All the atomic numbers from 1 to 92 were alreadyoccupied by known elements in 1913, except for seven - atomic numbers 43, 61,72, 75, 85, 87, and 91. In 1917, protactinium (atomic number 91) wasdiscovered. In 1923, hafnium (atomic number 72) was discovered, and in1925, rhenium (atomic number 75). Exactly four gaps were left then inthe periodic table: 43, 61, 85, and 87. Only four elements, it would seem,remained to be discovered; but those gaps remained well into the 1930's.
Since the proton is the only positively charged particle to be found in thenucleus, the atomic number is equal to the number of protons in the nucleus. Aluminum, with an atomic number of 13, has to contain 13 protons in its nucleus. But since its atomic weight is 27, it must also contain (as was laterdiscovered) 14 neutrons in its nucleus. The neutrons contribute to the mass butnot to the charge. In the same way, a sodium atom with an atomic number of 11and an atomic weight of 23 must have a nucleus with 11 protons and 12 neutrons.(Because protons and neutrons are both found in the nucleus, they are lumpedtogether as nucleons.)
The atom in its normal state is electrically neutral. This means that forevery proton in the nucleus there must be an electron in the outskirts.Therefore, the number of electrons in the neutral atom is equal to the atomicnumber. A hydrogen atom contains 1 electron, a sodium atom 11 electrons, auranium atom 92 electrons, and so on. (Of course, positive ions have lostelectrons and negative ions have gained them. A sodium ion, therefore, hasfewer electrons than its atomic number, while a chloride ion has more electronsthan its atomic number.)
Electron Shells
When two atoms collide and react, they either cling together, sharing anumber of electrons, or separate again after having transferred one or moreelectrons from one atom to the other. It is this sharing of transferring ofelectrons that results in the changes of property noted in the substancesundergoing chemical reactions.
A certain amount of order with respect to the manner in which such electronschanges occur began to appear from the careful work that was done with thecharacteristic x-rays. Out of this work arose the concept that the electrons inan atom existed in groups that might be pictured as electron shells. The shells can be visualized as enclosing the nucleus like the rings in anonion, each successive shell capable of holding more electrons than the oneswithin. The shells were lettered K, L, M, N, O, P, and Q.
The innermost shell, the K-shell, can hold only two electrons, the L-shellcan hold eight, the M-shell as many as eighteen, and so on. This concept servedto explain the periodic table.
The three electrons of the lithium atom are arranged 2,1 among the electronshells; the eleven electrons of the sodium atom are arranged 2,8,1; the nineteenelectrons of the potassium atoms are arranged 2,8,8,1; and so on. Each of thealkali metals has the electrons of its atoms so arranged that the outermostoccupied electron shell contains just one electron.

Since it is the outermost electron shell that makes contact in collisionsbetween atoms, it is the number of electrons in that outermost shell that wouldbe expected to determine the chemical activity of an element. Different elementswith the outermost electron shells similar would have related properties. It isfor this reason that the various alkali metals are so similar in theirproperties.
In the same way, the alkaline earth elements (magnesium, calcium, strontium,and barium) are all similar, for each possesses two electrons in the outermostshell. The halogens (fluorine, chlorine, bromine, and iodine) all possess sevenelectrons in the outermost shell; while the inert gases (neon, argon, krypton,and xenon) all possess eight.
Indeed, Mendeleev, in arranging his periodic table, had - without knowing it- placed the elements into rows and columns in accordance with the arrangementof their atoms among the electron shells.
As more and more electrons are to be found in the heavier atoms, theelectron shells begin to overlap. Atoms of successive atomic numbers have addedelectrons to an inner shell, but the number of electrons in the outermost shellhas remained constant. This configuration happens, in particular, with the rareearth elements, the atomic numbers of which range from 57 to 71 inclusive. While we find an increase in inner shell electrons as we go up the periodictable, all the rare earths retain three electrons in their outermost shell. That similarity of outermost shells explained, at last, why the elements of thisgroup were so unexpectedly similar in their properties.
Mendeleev had arranged his periodic table by considering the valence of thedifferent elements, rather than their electronic arrangements, which wereunknown to him. It seemed reasonable to suppose that the valence of an elementwas governed by its electronic arrangement.
The German chemist Richard Abegg (1869-1910) hadpointed out, in 1904, that the inert gases must have a particularly stableelectronic configuration. The inert gas atoms had no tendency to add to orsubtract from this number, and that was why they did not participate in chemicalreactions. It followed that other atoms might give up or accept electrons inorder to achieve the inert gas configuration.
Thus, sodium's eleven electrons are 2,8,1 while chlorine's seventeenelectrons are 2,8,7. If sodium gives up an electron and chlorine accepts one,the former achieves the 2,8 configuration of neon and the latter the 2,8,8configuration of argon.
The sodium atom, in giving up a negatively charged electron, is left with apositive charge and becomes the sodium ion. The chlorine atom in gaining anelectron gains a negative charge and becomes the chloride ion. The two tend tocling together by virtue of electric attraction between positive and negative,as Berzelius had suspected a century earlier.
It is clear, from this consideration, why sodium should have a valence of 1. It cannot give up more than one electron without breaking up the stable 2,8arrangement. Nor can the chlorine atom accept more than one electron. On theother hand, calcium, with a 2,8,8,2 arrangement, and thus a valence of 2, tendsto give up two electrons, and oxygen, with a 2,6 arrangement, tends to accepttwo electrons.
It is these electron shifts that make it possible to set up concentrationsof charge in one place or another, so that chemical reactions can serve as asource for electric current, as Volta had discovered over a century earlier.
From the electronic view, equivalent weight turned out to represent therelative weights of elements involved in a single electron shift of this sort. The equivalent weight is, after all, the atomic weight divided by the valenceor, in other words, the atomic weight divided by the number of electronstransferred.
Abegg's suggestion only considered complete transfers of electrons from oneatom to another, producing electrically charged ions which then held together byelectrostatic attraction. This is electrovalence. Two Americanchemists, Gilbert Newton Lewis (1875-1946) and IrvingLangmuir (1881-1957), independently extended this notionin the years following 1916. They suggested an explanation, for instance, forthe structure of the chlorine molecule, in which two chlorine atoms are tightlybound together. Surely, there is not reason for one chlorine atom to transferan electron to another chlorine atom, and surely they could not hold together byordinary electrostatic attraction. Both Berzelius's and Abegg's theories ofinteratomic attraction fall short in this situation.
The Lewis-Langmuir suggestion was, instead, that each atom could contributean electron to a shared pool. The two electrons in the shared pool remained inthe outermost electron shell of both atoms. The electron arrangement in thechlorine molecule might therefore by pictured as 2,8,6,(1:1),6,8,2 with bothshared electrons counting as part of the electron complement in each atom. Eachatom would then have the 2,8,8 configuration in place of the much less stable2,8,7 arrangement of the individual chlorine atoms. It is for that reason thatthe chlorine molecule is much more stable than are the free atoms.
In order to keep the electron pool in the outermost electron shell of bothatoms, the two atoms had to remain in contact, and it takes considerable energyto tear them apart. Each electron contributed to such a pool represents avalence of 1 for the atom doing the contributing. Such valence, requiring theaction of two atoms in cooperation, is covalence.
The Lewis-Langmuir theory was especially convenient for organic compounds,since the bonds between one carbon atom and another or between one carbon and ahydrogen atom were easily explained in this fashion. Most organic moleculescould therefore easily be represented by electronic formulas where theold dash of the Kekule formula was replaced by a shared electron pair.
In fact, the English chemist Nevil Vincent Sidgwick(1873-1952) was able, in the 1920's, to extend the notion of electron-paircovalence to inorganic compounds. In particular, he applied them to Werner'scoordination compounds where the ordinary Kekule representations were difficultto apply.
In all these chemical changes only electrons are being shifted. The protons(in all but one case) are safely protected in the central nucleus. Theexceptional case is that of hydrogen, which has a nucleus made up of a singleproton. If the hydrogen atom is ionized through removal of its single electron,the proton is left bare. (Such a bare proton is very active and does not remainbare for long. In water solution, it immediately attaches itself to a watermolecule, adding a positively charged hydrogen atom to that molecule. Thus isformed the hydronium ion, H3O+.)
In 1923, the Danish chemist Johannes Nicolaus Bronsted(1879-1947) introduced a new view of acids and bases. An acid was defined as acompound tending to give up a proton (or hydrogen ion), while a base was onetending to combine with a proton. This new view accounted for all the factsalready satisfactorily accounted for by the old view of Svante Arrhenius. Inaddition it represented a greater flexibility that made it possible to extendacid-base notions into areas in which the old view was inadequate.
The relatively small molecules and rapid, ionic reactions in inorganicchemistry had proven comparatively easy to study. Chemists, from Lavoisier'stime onward, could predict the course of such reactions and the manner ofmodifying them to suit needs. The complicated molecules and slow reactions inorganic chemistry were much harder to analyze. Often there were several ways inwhich two substances could react; guiding the reaction into some desired pathwas a matter of art and intuition rather than of secure knowledge.
The electronic atom offered organic chemists a new look at their field. Inthe late 1920's, men such as the English chemist Christopher Ingold(1893- ) began to try to interpret organic reactions in terms of electron shiftsfrom point to point within a molecule. The methods of physical chemistry beganto be applied intensively in an attempt to interpret the directions andtendencies of such shifts. Physical organic chemistry became animportant discipline.
In proved insufficient to attempt to interpret organic reactions in terms ofhard little electrons moving here and there, and it did not long remainnecessary to do so.
For the first quarter-century after the discovery of the electron, it wastaken for granted that the particle was a tiny, hard sphere. In 1923 LouisVictor de Broglie, Prince de Broglie, a French physicist(1892-1987), had presented theoretical reasons for considering electrons (andall other particles as well) to possess wave properties. Overlord™ download for mac. Before the end of the1920's this view had been confirmed by experiment.
Pauling (the first to suggest the helical shape of proteins and nucleicacids), developed methods, in the early 1930's, for taking into account the wavenature of electrons in considering organic reactions. He showed that theLewis-Langmuir electron pool could be interpreted as wave-interactions. Electron waves paired off in reinforcement, resonating with each other to form astabler situation in combination than in separation.
This theory of resonance was particularly useful in establishing thestructure of benzene, which had been puzzling in Kekule's day and which hadretained questionable points ever since. As usually drawn, the structure ofbenzene is that of a hexagon with alternating single and double bonds. By theLewis-Langmuir system, two-electron pools and four-electron pools alternated. Benzene lacked almost completely the characteristic properties of othercompounds which contained double bonds, or four-electron pools.
Pauling showed that if electrons were regarded as wave-forms, the individualelectrons need not be considered as occupying a single point, but could 'smearout' over a considerable area. The 'electron waves' could spreadout to take up far larger areas than a tiny 'billiard ball' electroncould be expected to take up. The tendency to 'smear' in this fashionwas accentuated if a molecule was quite flat and symmetrical.
The benzene molecule is flat and symmetrical, and Pauling showed that theelectrons 'smeared out' in such a fashion that all six carbon atoms ofthe benzene ring were found in equal fashion. The bonds connecting them couldnot be represented as either single bonds or double bonds, but as a kind ofparticularly stable average, or resonance hybrid, between the twoextremes.
Nuclear Atom Picture
Other points besides the structure of benzene were clarified by the theoryof resonance. For instance, the four electrons in the outermost shell of thecarbon atom are not all equivalent from the standpoint of energycharacteristics. It might have been assumed, then, that bonds of slightlydifferent type would be formed between a carbon atom and its neighbor, dependingon which of carbon's electrons was involved.
It could be shown, though, that the four electrons, as wave-forms,interacted and formed four 'average' bonds that were preciselyequivalent, and directed toward the apices of a tetrahedron. Thus, the Van'tHoff-Le Bel tetrahedral atom was explained in electronic terms.
Resonance also helped to explain a group of strange compounds that had firstimpinged on the chemical consciousness at the opening of the twentieth century. In 1900, the Russian-American chemist Moses Gomberg(1866-1947) was trying to prepare hexaphenylethane, a compound with a moleculeconsisting of two carbon atoms to which six benzene rings were attached (threeper carbon atom).
He obtained, instead, a colored solution of a very reactive compound. Forvarious reasons, he was forced to conclude that he had obtained triphenylmethyl,a 'half-molecule' consisting of a carbon atom with three benzene ringsattached. The fourth valence bond of the carbon atom remained unused. Such acompound resembled one of the old radicals torn loose from a molecule. It wastherefore termed a free radical.
Once the electronic atom was introduced, a free radical such astriphenylmethyl was understood to contain an unpaired electron in the placewhere the old Kekule view would have put an unused bond. Ordinarily, such anunpaired electron is highly unstable. However, if a molecule is flat and highlysymmetrical, as triphenylmethyl is, the unused electron can be 'smeared out'over the entire molecule. The free radical is then stabilized.
When organic reactions came to be studied in electronic terms, it becameclear that there were usually stages where a free radical had to be formed. Such free radicals, generally not stabilized by resonance, could exist onlymomentarily and could be formed only with difficulty. It was the difficulty offorming free radical intermediates that made the most organic reactions so slow.
In the second quarter of the twentieth century, organic chemists werebeginning to get considerable insight into the detailed steps that made uporganic reactions - the reaction mechanism, in other words. It was thisinsight, more than anything else, which has guided contemporary organic chemistsin their synthetic work and has led to the syntheses of molecules whosecomplexities had defeated earlier generations.
Nuclear Atom Physics
Nor were resonance considerations confined to organic chemistry alone. Theboron hydrides possessed molecules that could not be neatly represented by olderviews. The boron atom possessed too few valence bonds (or electrons) for thepurpose. Yet if the electrons were properly 'smeared' as wave forms,a reasonable molecular structure could be proposed.
Again, in 1932, Pauling reasoned that the inert gas atoms could not be asresistant to forming bonds as had been assumed for the third of a century thathad elapsed since their discovery. Under sufficient pressure by an extremelyreactive atom such as that of fluorine, compounds might be formed.
This suggestion of Pauling's went unheeded at first, but in 1962, xenonfluoride was formed by reacting the inert gas xenon with fluorine. In shortorder a number of xenon compounds with fluorine and with oxygen were formed, aswell as one or two of radon and of krypton.
If the studies of the internal atomic structure had led to new insights andunderstandings, they also posed a normal share of new problems.
In 1900, Crookes had discovered that freshly prepared pure uranium compoundswere only very slightly radioactive, but that their radioactivity strengthenedon standing. By 1902 Rutherford and a co-worker, the English chemist FrederickSoddy (1877-1956), proposed that as a uranium atom gave offan alpha particle, its nature changed. It became a new type of atom, withdifferent radioactive characteristics, producing stronger radiations thanuranium itself (thus accounting for Crooke's observations).
This second atom in turn broke down, forming still another type of atom. Indeed, the uranium atom was the parent of a whole series of radioactiveelements, a radioactive series, that included radium and polonium andended finally with lead, which was not radioactive. It was for this reason thatradium, polonium, and other rare radioactive elements could be found in uraniumminerals. A second radioactive series also began with uranium, while a thirdseries began with thorium.
(This breakdown of uranium into lead would, by Boyle's definition ofelements, have made it necessary to view uranium as not being an element. Bythe new atomic number definition it was still an element. It is just that sinceatoms are not really indivisible particles after all, elements are notnecessarily entirely unchangeable. This represents a return - on a much higherlevel of sophistication - to the old alchemical concept.
It is reasonable to ask why, though, if radioactive elements are constantlybreaking down, any remained in existence at all? It was Rutherford who, in1904, solved the matter. In studying the rate of radioactive breakdown, he wasable to show that after a certain period, which was different for each element,half of any given quantity of a certain radioactive element would have brokendown. This period, which is characteristic for each particular type ofradioactive substance, Rutherford called the half-life.
The half-life of radium is just under 1600 years. Over the geological erasany radium in the earth's crust would certainly have long since vanished, wereit not that new supplies are constantly being formed through the breakdown ofuranium. The same is true for other breakdown products of uranium, some ofwhich have half-lives of only fractions of a second.
The half-life of uranium is 4.5 X 109 years. This is atremendous period of time, and in all the history of the earth, only a fractionof the original supply of uranium has had a chance to break down. Thoriumbreaks down even more slowly, its half-life being 1.4 X 1010 years.
Such huge stretches of time can be determined by counting the number ofalpha particles produced by a given mass of uranium (or thorium). The alphaparticles were counted by Rutherford, by noting the small flashes they made whenthey struck a screen of zinc sulfide. (This was a scintillation counter.)
Each alpha particle given off meant a uranium atom breaking down so thatRutherford could determine how many atoms were breaking down per second. Fromthe mass of the uranium he was dealing with, he knew the total number of uraniumatoms present. With this information, he could easily calculate how long itwould take for half the uranium atoms present to break down, and it turned outto be a matter of billions of years.
So constant and characteristic is the majestically slow decay of uraniumthat it can be used to measure the age of the earth. In 1907, the Americanchemist Bertram Borden Boltwood (1870-1927) suggestedthat the lead content of uranium minerals would serve as guide in this respect. If it is assumed that all the lead in the mineral originated from uranium decay,it would be easy to calculate how long a time must have elapsed to bring thatamount of lead into existence. It was eventually calculated in this way thatthe solid crust of the earth must have been in existence for at least fourbillion years.
Meanwhile, Soddy had gone on to describe the exact manner in which an atomchanged as it gave off subatomic particles. If an atom lost an alpha particle,with a charge of +2, the total charge on its nucleus was decreased by two. Snakeybus for mac. Theatom moved two places to the left in the periodic table.
If an atom lost a beta particle (an electron with a charge of -1), thenucleus gained an additional positive charge (In Soddy's time, it was felt thatthere were electrons in the nucleus and that the loss of a beta particle fromthe nucleus left an additional proton unbalanced, hence raised the positivecharge. Nowadays, it is felt that the nucleus contains only protons andneutrons, but that an electron is formed and expelled when a neutron isconverted into a proton, for the gain of a positive charge is equivalent to theloss, by expulsion, of a negative charge.) and the element moved one place tothe right in the periodic table. If an atom lost a gamma ray (uncharged), itsenergy content was altered but there was no change in its particle makeup, sothat it remained the same element.
Using these rules as a guide, chemists could work out the details of thevarious radioactive series.
But all this raised a serious problem. What was one to do with the variousbreakdown products of uranium and thorium? Dozens of these were discovered, butthere were at most only nine places in the periodic table (from polonium atatomic number 84 to uranium at atomic number 92) in which to place them.
As a specific example, the uranium atom (atomic number 92) emitted an alphaparticle and the atomic number of what was left of the atom therefore became 90,by Soddy's rule. This meant that a thorium atom had been formed. However,whereas ordinary thorium had a half-life of 14 billion years, the thoriumproduced from uranium had a half-life of 24 days.
Differences existed even in the case of non-radioactive elements and inproperties not involving radioactivity. For instance, Richards (the expert onatomic weights) was able to show, in 1913, that the lead produced by the decayof uranium did not have quite the same atomic weight as ordinary lead.
Soddy advanced the bold suggestion that more than one kind of atom could fitinto the same place in the periodic table. Place number 90 might hold differentvarieties of thorium, place number 82 different varieties of lead, and so on. He called these atom-varieties occupying the same place isotopes, fromthe Greek word meaning 'same place'.
The different isotopes in a given place in the table would have the sameatomic number, therefore the same number of protons in the nucleus and the samenumber of electrons in the outskirts. The isotopes of an element would have thesame chemical properties, since these properties depend on the number andarrangement of the electrons in the atoms.
But in that case, how does one explain differences in radioactive propertiesand in atomic weight?
Atomic weight might represent the key to the difference. A hundred yearsearlier, Prout had advanced his famous hypothesis that all atoms are composed ofhydrogen so that all elements should have integral atomic weight. The fact thatmost atomic weights are not integers seemed to have destroyed his hypothesis.
But now the atom, in its new nuclear guise, had to be made up of protons(and neutrons). Protons and neutrons are about equally massive, and therefore,all atoms had to have weights that were integral multiples of the weight ofhydrogen (made up of a single proton). Prout's hypothesis was reinstated, and alook of new suspicion was directed at the atomic weights instead.
In 1912, J. J. Thomson (the discoverer of the electron) had subjected beamsof positively charged neon ions to the action of a magnetic field. The fielddeflected the neon ions and caused them to fall on a photographic plate. If allthe ions had been identical in mass they would all have been deflected by thesame amount, and a single discolored spot on the photographic film would haveappeared. However, two spots were located, one some ten times as dark as theother. A co-worker, Francis William Aston (1877-1945),later improved the device and confirmed the results. Similar results wereuncovered for other elements. Since this device separated chemically similarions into a kind of spectrum of dark spots, it was called the massspectrograph.
The extent of deflection of ions of identical charge by a magnetic fielddepends upon the mass of the ion; the more massive the ion, the less it isdeflected. From the results obtained by Thomson and Aston it would seem thatthere were two kinds of neon atoms, one more massive than the other. One typehad a mass number of 20 and the other, a mass of 22. Since the neon-20was ten times as common as neon-22, judging from the relative darkness of thespots (in later years tiny quantities of neon-21 were also isolated), it wasreasonable that the atomic weight of neon was about 20.2.
In other words, individual atoms had masses that were an integral multipleof that of the hydrogen atom, (Not quite a multiple, in actual fact. The smalldeviations in mass are of no importance in chemistry but are a reflection of thehuge energies involved in nuclear forces - energies that have been made manifestin nuclear bombs.) but a particular element, being made up of atoms of differentmass, would have an atomic weight that was a weighted average of these integersand would therefore not necessarily be an integer itself.
Structure Of The Nuclear Atom
The weighted average of the isotopes of a particular atom may be greater, insome cases, than the weighted average for an atom of higher atomic number.
For instance, tellurium, with an atomic number of 52, consists of sevenisotopes. Of these, the two most massive isotopes, tellurium-126 andtellurium-128, are the most common. The atomic weight of tellurium thereforecomes to 127.6. Iodine has the next higher atomic number, 53, but it is made upof iodine-127 only and therefore has the atomic weight of 127. When Mendeleevplaced iodine after tellurium in his periodic table, reversing the orderdictated by atomic weight, he was, without knowing it, following atomic numberinstead; and this was the correct thing to do.
Nuclear Atom Splitting
Here's another example. Potassium (atomic number 19) is made up of threeisotopes, potassium-39, potassium-40, and potassium-41, but the lightestisotope, potassium-39, is by far the most common. Hence, the atomic weight ofpotassium is 39.1. Argon has a lower atomic number (18) and is made up of threeisotopes also, argon-36, argon-38, and argon-40. Here, however, it is the mostmassive isotope, argon-40, which is most common. Therefore the atomic weight ofargon is about 40. When Ramsay placed argon before potassium instead of afterin defiance of atomic weights, he, too, without knowing it, was following atomicnumber and was doing the correct thing.
The use of the mass spectrograph made it possible to determine atomic weightby actually measuring the mass of the individual isotopes and the quantity ofeach present - and then taking the average. This method surpassed chemicalmethods for measuring atomic weight in accuracy.
Different isotopes of a given element have the same atomic number butdifferent mass numbers. The different isotopes would have the same number ofprotons in their nucleus but different numbers of neutrons. Thus, neon-20,neon-21, and neon-22 all have 10 protons in the nucleus, so that all have anatomic number 10, and all have an electron arrangement of 2,8. However, neon-20has a nucleus containing 10 protons plus 10 neutrons; neon-21, one containing 10protons plus 11 neutrons; and neon-22, one containing 10 protons plus 12neutrons.
Nuclear Atom Curtains Panels
Most elements (but not all) could be divided into isotopes in this manner. In 1935, the Canadian-American physicist Arthur Jeffrey Dempster(1886-1950) found that uranium, as it occurred in nature, was a mixture of twoisotopes even though its atomic weight (238.07) was close to a whole number. Itwas just that one isotope existed in overwhelming proportion. Fully 99.3 percent of the uranium atoms had nuclei made up of 92 protons and 146 neutrons or atotal mass number of 238. These were uranium-238 atoms. The remaining 0.7 percent had three fewer neutrons and were uranium-235 atoms.
Since radioactive properties depend upon the constitution of the atomicnucleus, and not upon electron arrangement, the isotopes of an element might besimilar chemically, but quite different from the standpoint of radioactivity. Thus, whereas uranium-238 has a half-life of 4.5 X 1010 years, thatof uranium-235 was only 7.0 X 108 years. (This accounts also for thedifference, mentioned earlier in the half-lives of natural thorium (thorium-232)and the thorium formed from the breakdown of uranium (thorium-234) whichcontains two additional neutrons in each nucleus.) Both are parents of separateradioactive series as well.
There were theoretical reasons for suspecting that hydrogen itself, thesimplest element, might be made up of a pair of isotopes. Ordinary hydrogenatoms, with nuclei composed of a single proton, make up hydrogen-1. In 1931 theAmerican chemist Harold Clayton Urey (1893-1981) slowlyevaporated four liters of liquid hydrogen on the presumption that if any heavierisotope of hydrogen existed, it would have a higher boiling point and wouldevaporate more slowly. This meant it would remain behind and accumulate in theresidue.
Sure enough, in the final cubic centimeter of hydrogen Urey was able todetect unmistakable signs of the existence of hydrogen-2, the nucleus of whichconsisted of one proton plus one neutron. Hydrogen-2 received the special nameof deuterium.
Nor was oxygen immune. In 1929, the American chemist William FrancisGiauque (1895-1982) succeeded in showing that oxygen wasmade up of three isotopes. The most common variety, comprising nearly 99.8 percent of all the atoms, was oxygen-16. It nucleus contained 8 protons plus 8neutrons. The rest were almost all oxygen-18 (8 protons plus 10 neutrons) witha trace of oxygen-17 (8 protons plus 9 neutrons).
This created a problem. Ever since the days of Berzelius, the atomicweights had been based on the arbitrary assignment of a weight of 16.0000 to theoxygen atom. But the atomic weight of oxygen could be only the weighted averageof the three isotopes, and the proportion of the isotopes in oxygen might varyslightly from sample to sample.
The physicists took to determining atomic weights on the basis of oxygen-16set equal to 16.0000, and this gave them a series of values (the physicalatomic weight) that were uniformly greater, by a very small amount, than thevalues that had been used and gradually improved throughout the nineteenthcentury (the chemical atomic weights).
In 1961, however, international organizations of both chemists and ofphysicists agreed to adopt an atomic weight standard based on carbon-12 setequal to exactly 12.0000. This new standard was almost exactly that of the oldchemical atomic weights and yet it was tied to a single isotope and not to theaverage of a group of them.
